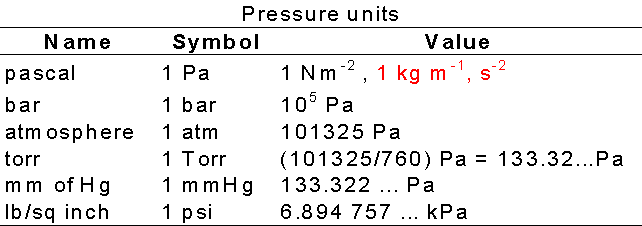
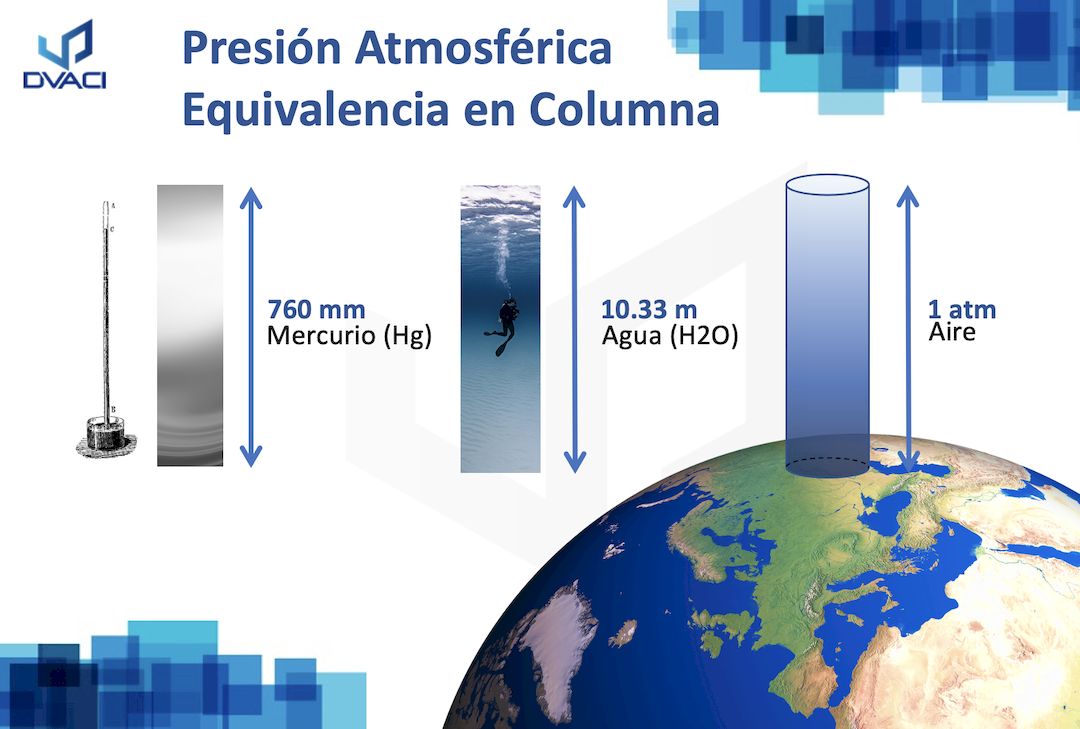
Volume of 0.5 mole of a gas 1 atm. pressure and 273 K is 22.4 litres11.2 litres5.6 litres44.8 litres
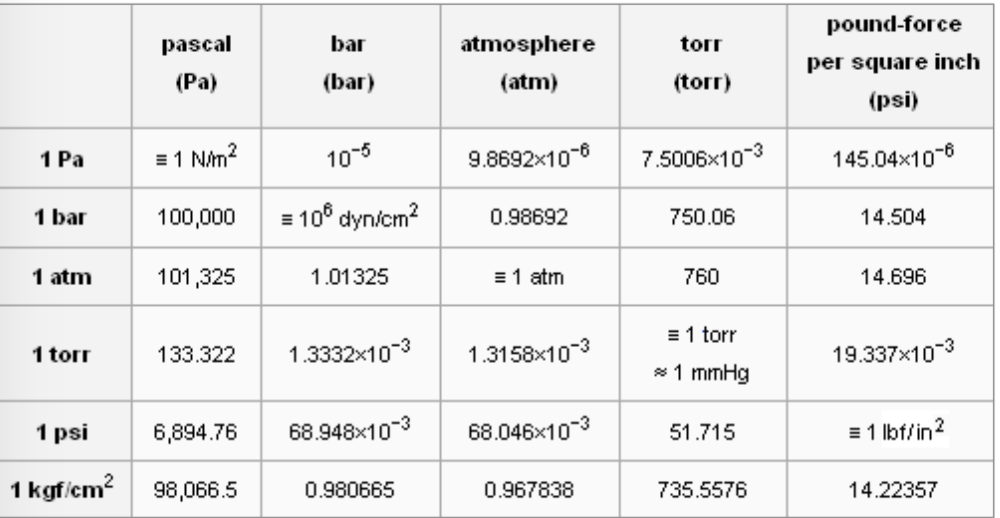
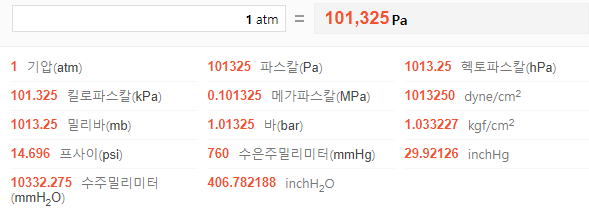
![SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1 SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1](https://cdn.numerade.com/ask_images/a1b97c8ec9ce48bcb5b41f914204e5b4.jpg)
SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1

Diagramme X-Y pour le mélange réactif à P = 1 atm. X-Y diagram for the... | Download Scientific Diagram

3 Diagrammes de phases binaire du système Si-C calculé à 1 atm ( a) et... | Download Scientific Diagram

At 100^oC and 1 atm pressure the density of water vapour is 0.0005970 g/cc.What is the molar volume and how does this compare with ideal gas volume ?What is the compressibilty factor '
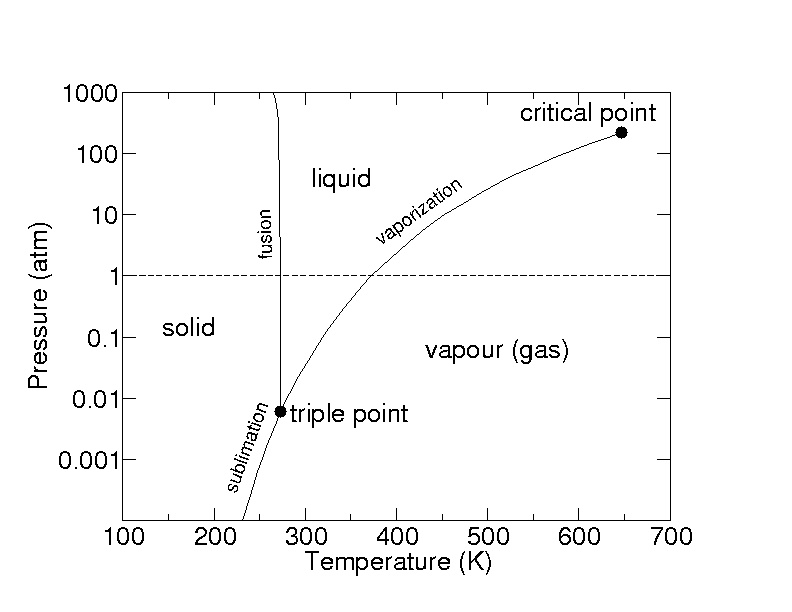
2.5: A pressure-temperature diagram (phase diagram) for water. Water at 1 atm pressure is on the dotted line

Déterminer l'état physique de l'eau à l'aide de sa température, sa pression et du diagramme d'état de l'eau - Tle - Exercice Enseignement scientifique - Kartable